Preclinical Validation of [177Lu]Lu-AKIR001, a CD44v6-Targeted Radiotherapeutic Entering First-in-Human Trials
2025.11.06.
Anja C L Mortensen et al., The Journal of Nuclear Medicine, 2026
Abstract
Targeted radionuclide therapy is an emerging potent therapeutic strategy in oncology. The cell surface antigen CD44v6 is a potential pan-cancer target for radionuclide therapy. This study aimed to evaluate the therapeutic efficacy, biodistribution, dosimetry, and safety profile of AKIR001, an antibody targeting CD44v6 labeled with 177Lu. Methods: The biodistribution and preclinical dosimetry of [177Lu]Lu-AKIR001 were calculated in the highly CD44v6-expressing A431 murine xenograft model, with subsequent extrapolation to predict human dosimetry. Therapeutic efficacy was evaluated across 3 xenograft models, 2 with high and 1 with moderate levels of CD44v6, using multiple dosing levels, fractionation regimens, and combinations with cisplatin. Preclinical toxicology was evaluated in a cross-reactive rabbit model and complemented by a PET imaging study using 68Ga-labeled AKIR001 in a cynomolgus macaque. Results: Biodistribution studies confirmed the high and selective tumor uptake of [177Lu]Lu-AKIR001, resulting in favorable dosimetry predictions for clinical application. Therapeutic evaluations demonstrated significant dose-dependent efficacy in all tested xenograft models, with fractionated dosing (2 doses) resulting in complete tumor regression in 80% of the animals in a radioresistant xenograft model. Biodistribution in rabbits demonstrated low uptake in normal tissues, and a good-laboratory-practice study using an excessive dose of AKIR001 was well tolerated, with no signs of adverse effects. PET imaging in a cynomolgus macaque corroborated these findings. Conclusion: Collectively, these data strongly support the therapeutic efficacy, safety, and dosimetry of [177Lu]Lu-AKIR001, justifying its advancement into clinical trials. A phase 1 clinical trial of [177Lu]Lu-AKIR001for CD44v6-positive solid cancers (NCT06639191) is currently recruiting patients.
Results from nanoScan® SPECT/CT and MultiScan™ PET/CT
-
Biodistribution studies were performed in BALB/c nu/nu mice carrying A431 xenografts (high CD44v6 expression). Increasing tumor uptake was observed, peaking at 65 %ID/g at 96 h after injection. Blocking with an excess of unlabeled AKIR001 reduced uptake to 19 %ID/g (P < 0.0001), confirming specificity.
Figure 1.: Biodistribution and small-animal SPECT imaging at 3 dose levels of 177Lu-AKIR001 in A431 xenografts. (A) Biodistribution of 177Lu-AKIR001 (15 µg) in BALB/c nu/nu mice carrying A431 xenografts, terminated at 4, 24, 48, 96, 168, and 240 h after injection. In vivo block using 30-fold molar excess of nonradiolabeled AKIR001 was evaluated at 96 h after injection.
- Dose-Dependent and Target-Specific Effect of 177Lu-AKIR001
 Figure 3.: Efficacy studies on ACT-1. (A) Kaplan–Meier survival of animals carrying ACT-1 xenografts (high CD44v6) treated with phosphate-buffered saline, nonradiolabeled AKIR001, [177Lu]Lu-iso control (AK-MO176-156, hIgG1 LALA isotype control, 17 MBq), or high dose (17 MBq) or low dose (8 MBq) of [177Lu]Lu-AKIR001. (B) Small-animal SPECT of mouse injected with 17 MBq of [177Lu]Lu-AKIR001 at 4 d after injection. (C) Individual tumor growth by treatment group.
Figure 3.: Efficacy studies on ACT-1. (A) Kaplan–Meier survival of animals carrying ACT-1 xenografts (high CD44v6) treated with phosphate-buffered saline, nonradiolabeled AKIR001, [177Lu]Lu-iso control (AK-MO176-156, hIgG1 LALA isotype control, 17 MBq), or high dose (17 MBq) or low dose (8 MBq) of [177Lu]Lu-AKIR001. (B) Small-animal SPECT of mouse injected with 17 MBq of [177Lu]Lu-AKIR001 at 4 d after injection. (C) Individual tumor growth by treatment group.
-
To evaluate [177Lu]Lu-AKIR001 against lower-expressing tumors, efficacy was evaluated in BHT-101 xenografts with moderate CD44v6 expression levels.
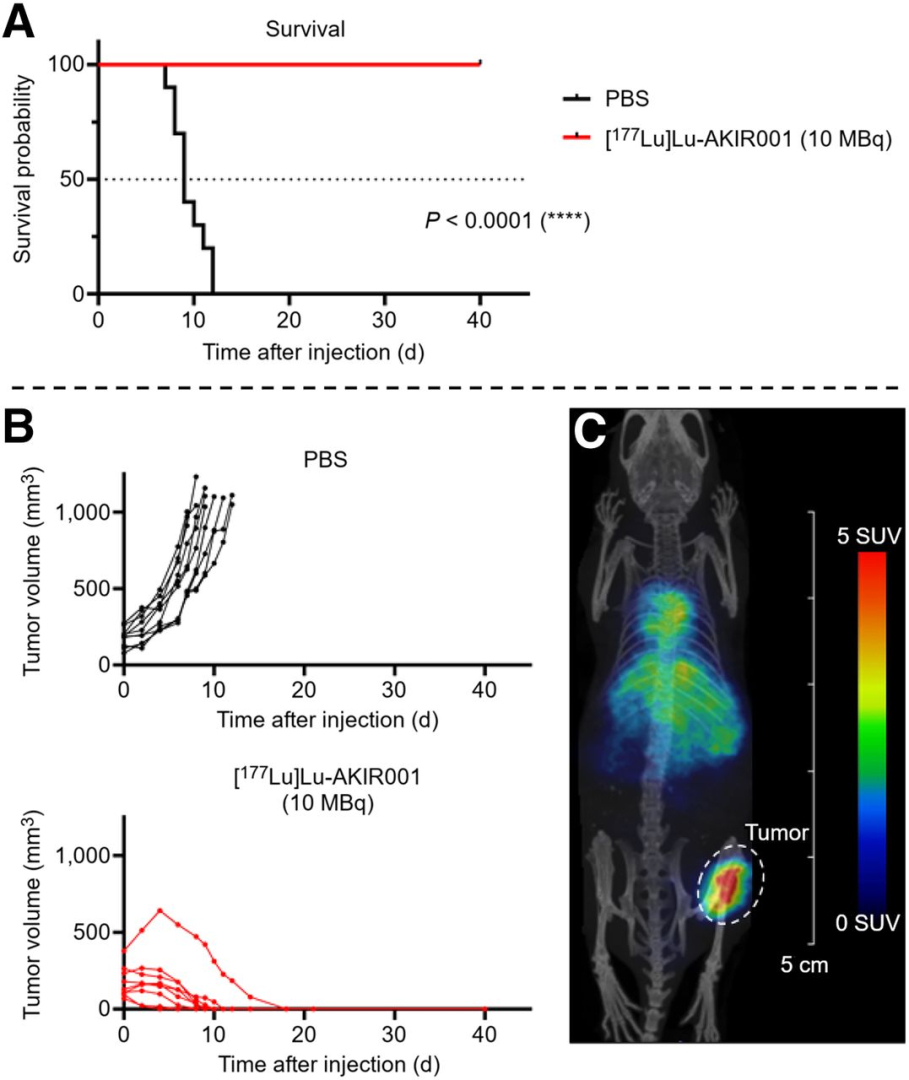
Figure 4.: Efficacy studies on BHT-101. (A) Kaplan–Meier survival plot of mice carrying BHT-101 xenografts treated with either placebo (phosphate-buffered saline, n = 10) or 10 MBq of [177Lu]Lu-AKIR001 (n = 9, N = 19). (B) Individual tumor growth by treatment group. (C) Small-animal SPECT/CT of mouse carrying BHT-101 xenograft injected with 10 MBq of [177Lu]Lu-AKIR001 at 4 d after injection.
-
AKIR001’s cross-reactivity with rabbit CD44v6 enabled biodistribution studies on New Zealand white rabbits using [111In]In-AKIR001 (>90% labeling yield), and to assess potential off-target binding in normal tissues, a [68Ga]Ga-labeled UU-40 imaging study was conducted on a cynomolgus monkey.
 Figure 5.: Biodistribution of [111In]In-AKIR001 in male (A) and female (B) New Zealand white rabbits (presented as percentage injected dose [%ID] per gram) at 4, 24, 72, and 168 h after injection (N = 9; n = 5 males and n = 4 females). (C) Whole-body 68Ga PET images of cynomolgus macaque showing biodistribution of [68Ga]Ga-UU-40IAHA at different time points.
Figure 5.: Biodistribution of [111In]In-AKIR001 in male (A) and female (B) New Zealand white rabbits (presented as percentage injected dose [%ID] per gram) at 4, 24, 72, and 168 h after injection (N = 9; n = 5 males and n = 4 females). (C) Whole-body 68Ga PET images of cynomolgus macaque showing biodistribution of [68Ga]Ga-UU-40IAHA at different time points.
Conclusion
[177Lu]Lu-AKIR001 is a CD44v6-targeted RPT with strong preclinical efficacy, specificity, and safety. Its broad tumor applicability and its potential for use in combination support clinical evaluation, and it is currently being assessed in a phase 1 trial (NCT06639191) for safety and therapeutic potential in CD44v6-expressing cancers.
Full article on jnm.snmjournals.org
How can we help you?
Don't hesitate to contact us for technical information or to find out more about our products and services.
Get in touch